 The valence of an element is the number of electrons the elements gain or loses when it forms compounds with other elements. Valence is the property related to the electrons in an atom’s outer shell. When atoms combine they gain, lose or share electrons in such a way that the outer shells become chemically complete. When atoms combine, they may gain, lose, or share:? There are molecules of compounds (eg CH4) and molecules of elements (eg O2). The atoms always join in fixed ratios and molecules have a specific formula, eg H2O or N2. 
The atoms in a molecule are joined by covalent bonds. How are atoms in a molecule chemically bonded?Ī cluster of non-metal atoms that are chemically bonded together. For example, nitrogen gas molecules are just two One may also ask, why are atoms combined? The atoms in a molecule can the the same. What is it called when atoms join together?Īnswer and Explanation: When atoms join together, the group is called a molecule. A water molecule has three atoms: two hydrogen (H) atoms and one oxygen (O) atom. An atom is the smallest particle of an element, like oxygen or hydrogen. It is possible for the same kinds of atoms to combine in different but definite proportions to form different molecules for example, two atoms of hydrogen will chemically bond with one atom of oxygen to yield a water molecule, whereas two atoms of hydrogen can chemically bond with two atoms of oxygen to form a … What atoms are water made up of?Įverything is made of atoms. In the kinetic theory of gases, the term molecule is often used for any gaseous particle regardless of its composition. Molecules are distinguished from ions by their lack of electrical charge. Is it true that molecule is composed of atoms Why?Ī molecule is an electrically neutral group of two or more atoms held together by chemical bonds. Why do atoms combine together and form molecules?Ītoms combine to form a molecule in order to achieve a stable configuration like those of the noble gases. When two atoms share electrons between them, they are locked together (bonded) by that sharing. Electrons can join (or bond) atoms together in two main ways. How do atoms join together to form molecules?Ītoms come together to form molecules because of their electrons. 5 What kind of bond is formed between oxygen and hydrogen?.4 How are atoms in a molecule chemically bonded?.2 Is it true that molecule is composed of atoms Why?.1 How do atoms join together to form molecules?.REVIEW: Atoms share electrons unequally in a (an) _ bond. REVIEW:A salt will dissolve in water to form REVIEW: What is formed when an atom loses or gains an electron? 
Finish off with the Conclusion and the Quiz. To get to another animation, click on the Options button. Read the Introduction, then click on Animation. This website has a good review of chemical bonding. This animation (Audio - Important) is a good review of the types of bonds. These bonds impart structure to liquid water and stabilize nucleic acids and other large molecules. Hydrogen Bonding: In a hydrogen bond, an atom of a molecule interacts weakly with a hydrogenatom already taking part in a polar covalent bond. Water is an example of a polar covalent bond. 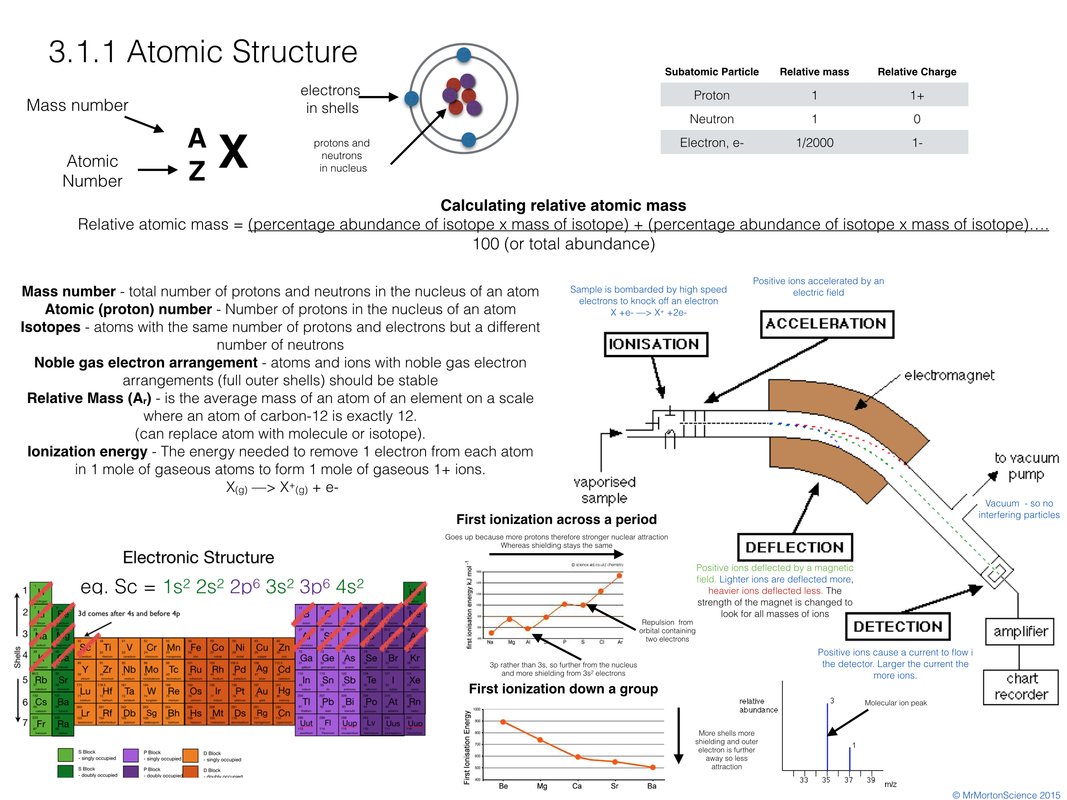
In a polar covalent bond, because atoms share the electron unequally, there is a slight difference in charge between the two poles of the bond. In a nonpolar covalent bond, atoms share electrons equally. The oppositely charged ions thenattract each other, forming an ionic bond.Ĭovalent Bonding: A covalent bond holds together two atoms that share one or more pairs of electrons. When chlorine accepts the electron, it becomes negatively charged. Sodium becomes positively charged when it gives up an electron to chlorine. The chlorine atom has seven electronsin the same shell. The sodium atom has only one electron in its outermost shell. Table salt ,composed of bonding between sodium (Na ) and chlorine (Cl-) ions is an example of an ionic bond. In an ionic bond, positive ( ) and negative (-) ions arelinked by mutual attraction of opposite charges. Ionic Bonding: When an atom loses or gains one or more electrons, it becomes positively or negatively charged and is called an ion. How atoms join together to form molecules How Atoms Join Together to Form Molecules There are three important types of bonds in biological molecules that we must learn: ionic, covalentand hydrogen. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
